Equipment: Brilouin Scattering Microscope (BSM)
Institution: Vienna Biocenter Core Facilities GmbH (VBCF)
Description: Our Brilouin Scattering Microscope (BSM) is a confocal-scanning implementation of a spectroscopy technique -Brillouin Scattering spectroscopy, which allows for all-optical label-free spatial mapping of the viscoelastic properties of materials. To measure the Brillouin Spectrum one probes the sample with a single-frequency laser and measures the GHz-scale (<0.0001nm wavelength) spectral modification scattered light.

This spectrally-shifted light – typically measured in the back-scattering configuration, is the result of light scattering from inherent collective thermal density fluctuations with the sample, and can be used to calculate the hypersonic velocity in the probed region. Given knowledge of the density and effective refractive index of the probed region one can then calculate the viscoelasticity of the sample, specifically the bulk longitudinal storage and loss moduli – in the GHz regime. Our setup allows for the combination with fluorescence or Raman scattering detection such that one can correlate the viscoelasticity and fluorescence or Raman spectra on a pixel-per-pixel level. The required acquisition times in order to get reasonable estimates of the viscoelasticity are on the order of several seconds per pixel for typical samples. We can achieve a spatial resolution down to ~300nm laterally, and can produce 3d viscoelasticity for reasonably transparent samples. The technique is starting to find increasing applications in medical diagnostics and biomedical research. For more information see here.
Equipment: Micro-Brillouin imaging; Raman microscopy; in vivo Raman probes; deep-Raman spectroscopy; micro-FTIR spectroscopic imaging
Institution: University of Exeter, UK

Description: A novel Brillouin microscope based on a dual-stage virtually imaged phase array (VIPA) spectrometer has been developed (Dr Palombo) as an advanced mechanical microscopy tool for biomedical samples. Several extracellular matrices and tissue types are being investigated in this regard using a mapping set-up with an inverted Olympus microscope, motorized PI stages and Andor sCMOS camera. Ongoing projects are aimed to test various optical approaches in combination with Raman spectroscopy for both normal tissue and pathology screening, and to answer fundamental questions on tissue biomechanics which can elucidate new targets for therapy. We are also developing a new microscope system based on NIR laser to enable improved match with spectral pathology based on Raman scattering. The BioSpec labs also host other advanced biophotonics facilities which are being applied or developed for clinical translation (cancer and dementia research primarily). These include Raman microscopes; in vivo Raman probes; deep-Raman systems; micro-IR and FTIR spectroscopic imaging; multivariate statistical analysis.
Equipment: Simultaneous Brillouin-Raman Microspectroscopy
Institution: Department of Physics and Geology, University of Perugia (IT)
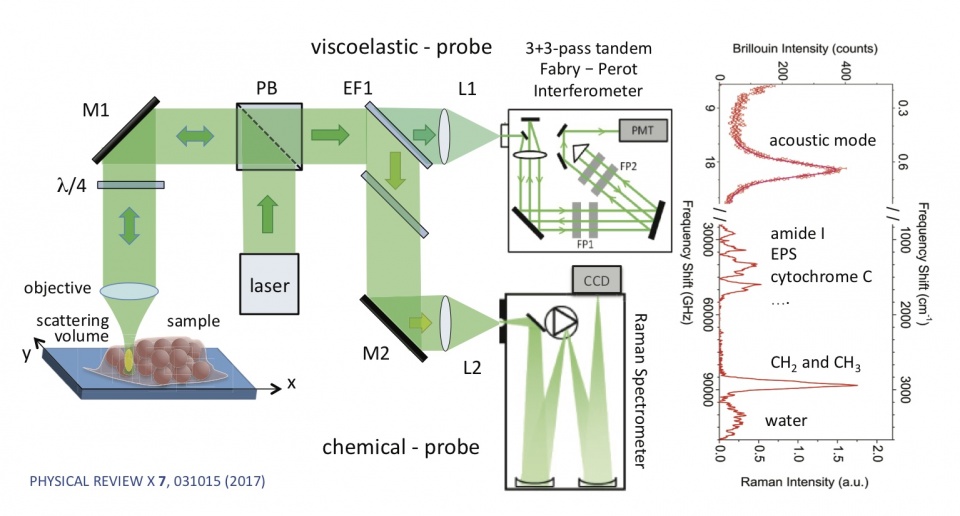
Description: High-resolution, high-contrast, and wide spectral range confocal microspectroscopic setup based on a new concept of multipass Fabry-Perot interferometer and a conventional Raman spectrometer, enabling simultaneous Brillouin-Raman microspectroscopy (BRMS) in an exceptionally wide spectral range, from fractions of GHz to hundreds of THz. It yields an unprecedented 150-dB contrast, which is especially important for the analysis of opaque or turbid media such as biomedical samples, and spatial resolution on a subcellular scale.
Equipment: Confocal scanning Brillouin scattering microscope (cs-BSM)
Institution: Heidelberg University
Description: Currently, we are in the process of establishing a cs-BSM system allowing parallel high resolution wide field imaging – in particular Differential Interference Contrast (DIC) and epi-fluorescence. The parallel detection of epi-fluorescence facilitates the identification of regions of interest and correlate morphological and biochemical properties with the probed elasticity. In cooperation with the Kareem Elayad (VBC, Vienna) such a system has allowed already the determination of the mechanical properties of probes from plant cell walls, our preferred research target, demonstrating its effectiveness to dissect mechanical aspects of plant morphogenesis.
Equipment: Custom built dual-stage VIPA confocal microscope (+488nm laser, incubator, Lyot stop)
Institution: European Molecular Biology Laboratory (EMBL)

Description: Based on inverted Zeiss 200, 532 Torus Brillouin laser, PMT confocal fluorescence detection (GFP band)
Equipment: AFM (CellHesion 200 from JPK/Bruker)
Institution: European Molecular Biology Laboratory (EMBL)
Equipment: Microscope at ICMM-CSIC
Institution: Instituto de Ciencia de Materiales de Madrid (ICMM-CSIC) (ES)
Description:
- Brillouin scattering measured: [text BM-type]
- Type of spectrometer: Sandercock 3+3 Tandem
- Spectral resolution and spectral range: FSR variable.
- Scan range: x,y (manual, several cm) no z scan
- Scan resolution: 1 micron
- Optimized for these types of samples: Brain and heart tissues.
Contact person: Dr. Rafael J. Jiménez Riobóo, rjimenez@icmm.csic.es